Translating…
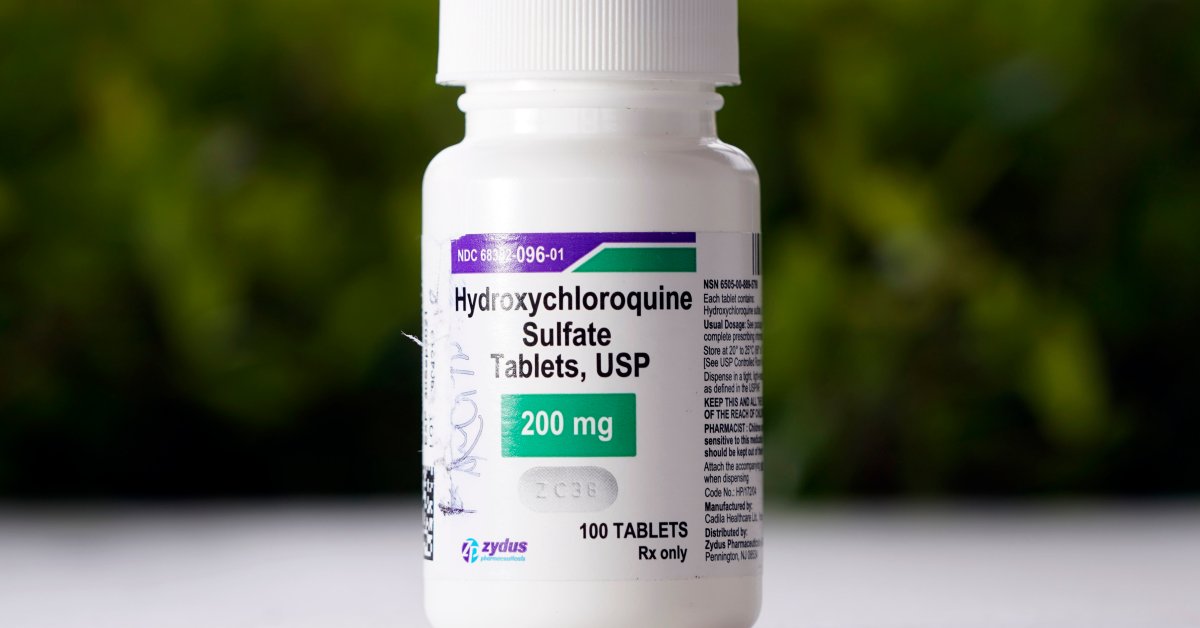
(GENEVA) — The World Successfully being Group talked about Monday that this can temporarily tumble hydroxychloroquine — the anti-malarial drug U.S. President Trump says he is taking — from its international glance into experimental COVID-19 treatments, asserting that its experts ought to verify all on hand proof up to now.
In a press briefing, WHO director-overall Tedros Adhanom Ghebreyesus talked about that in light of a paper revealed closing week in the Lancet that confirmed folks taking hydroxychloroquine were at better risk of demise and heart problems, there would be “a non everlasting pause” on the hydroxychloroquine arm of its international scientific trial.
“This disclose pertains to the usage of hydroxychloroquine and chloroquine in COVID-19,” Tedros talked about, including that the medicine are licensed treatments for folks with malaria or autoimmune diseases. Diversified treatments in the trial, including the experimental drug remdesivir and an HIV mixture therapy, are light being examined.
Tedros talked about the government crew at the help of WHO’s international “Solidarity” trial met on Saturday and determined to conduct a total overview of all on hand records on hydroxychloroquine and that its use in the trial would be suspended for now.
Dr. Michael Ryan, WHO’s emergencies chief, talked about there became once no indication of any safety problems with hydroxychloroquine in the WHO trial up to now, nonetheless that statisticians would now analyze the knowledge.
“We’re perfect performing on an abundance of caution essentially based totally on the recent outcomes of the entire studies to to make certain that we are able to proceed safely with that arm of the trial,” he talked about. WHO talked about it expected to have more information interior the following two weeks.
Final week, Trump announced he became once taking hydroxychloroquine even though he has not examined sure for COVID-19. His have administration has warned the drug can have deadly facet effects, and both the European Medicines Company and the U.S. Food and Drug Administration warned neatly being mavens closing month that the drug ought to not be archaic to treat COVID-19 delivery air of clinical institution or research settings attributable to a huge kind of extreme facet effects that in some cases could even be fatal.
Hydroxychloroquine and chloroquine are licensed for treating lupus and rheumatoid arthritis and for combating and treating malaria, nonetheless no huge rigorous checks have found them protected or efficient for combating or treating COVID-19.
___
Maria Cheng reported from London.
The Coronavirus Immediate.All the pieces it’s essential to know relating to the international unfold of COVID-19

Thank you!
To your safety, now we have despatched a affirmation electronic mail to the address you entered. Click the link to substantiate your subscription and delivery receiving our newsletters. Must you don’t acquire the affirmation interior 10 minutes, please verify your unsolicited mail folder.
Contact usateditors@time.com.